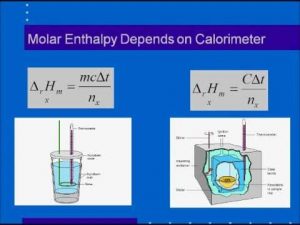
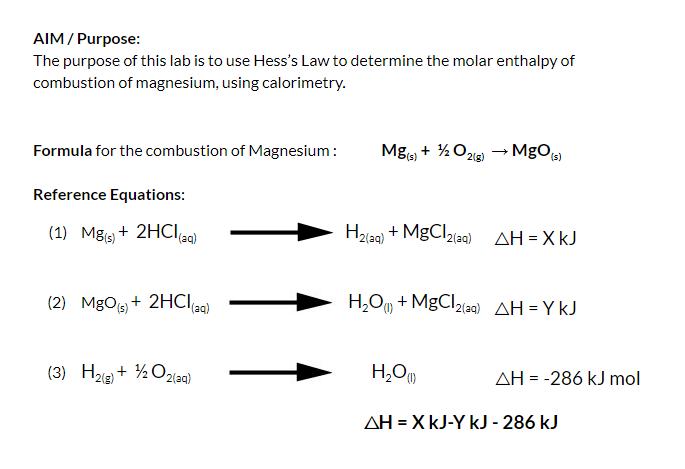
SOLVED:The molar enthalpy change for the neutralization reaction: HCI (aq) + NaOH (aq) +> NaCl (aq) + HzO (I) is AH=-57.3 k/mol. This is an exothermic reaction which means heat is released.
Experiment: To measure the molar enthalpy change for the dissolution ( enthalpy of solution) for vari - Tutorke